Demystifying Hydrogen
The Bottom Line
Hydrogen has the potential to eliminate 60 gigatons of CO2 emissions cumulatively from 2021-2050, according to the International Energy Agency. And according to McKinsey, the future "hydrogen economy" could create 700,000 jobs and $140 billion in annual revenue by 2030.
For reference: in 2019, the world emitted 51 gigatons of CO2-equivalent greenhouse gases. Project Drawdown estimates we need to cumulatively eliminate 1,000 GT from 2020-2050 to keep global warming below 2 degrees Celsius.
You Might Be Interested If...
You get energized by knotty problems and you want to tackle hard-to-abate sectors like aviation and steel
You're a high-risk, high-reward type and are intrigued by nascent energy technologies with transformative potential
You have a chemistry or physics degree, or you're comfortable speaking the language of molecules and electrons
What You Should Know
Source: Clean Energy Wire (https://www.cleanenergywire.org/news/germany-bets-green-hydrogen-quest-climate-neutrality)
Hydrogen, like electricity, is an incredibly adaptive "energy carrier," able to be used in everything from fuel refining to shipping to building heating to steel manufacturing. But using hydrogen is kind of like getting an MBA: there's cases where it makes total sense, there's cases where it's debatable, and there's cases where hey, maybe you should just stay in tech and pile up those stock options. (We just got our first tuition bill. It hurts.)
Let's start with the latter. There's been a lot of fanfare around hydrogen's ability to compete with or even replace electricity, but researchers like Stanford professor Mark Z. Jacobson caution that this is far from the truth. In popular applications like [passenger cars](https://cleantechnica.com/2021/02/01/chart-why-battery-electric-vehicles-beat-hydrogen-electric-vehicles-without-breaking-a-sweat/](https://cleantechnica.com/2021/02/01/chart-why-battery-electric-vehicles-beat-hydrogen-electric-vehicles-without-breaking-a-sweat/)) or heating, hydrogen is simply the less efficient option — as we'll discuss later, clean hydrogen is created using electricity, begging the question of why we wouldn't just use that electricity in the first place and save the energy lost in the conversion process.
Turns out that there is one great reason: hydrogen is light, even in large amounts (recall from high school chemistry that it's the tiniest element on the periodic table!). This lightness is a crucial factor for long-haul, heavy-duty transportation like trucks, ships, and airplanes, which require a lot of power to move. Powerful electric batteries are very heavy, which means the vehicle must exert more energy to carry them, which requires a larger battery, which means more weight, which requires more energy, etc. Because of this, by 2030, hydrogen fuel cell vehicles are expected to be the most cost-effective option for certain types of buses, trains, taxis, and trucks, and a cost-effective option compared to other low-carbon options for shipping and aviation.
The small size of hydrogen also means a large amount of it can be contained in a relatively small space, making it a potential solution for one of the central problems of climate tech: energy storage. Using renewable sources like solar and wind to power electric grids has a huge positive impact on emissions, but solar and wind are inherently inconsistent (or "intermittent") because of things like, you know, seasons. To smooth out this intermittency, electricity can be used to produce hydrogen via an electrolyzer in times of excess (e.g. sunny days), and, in later down times (e.g. winter), the hydrogen can be fed into a fuel cell to create electricity.
But again, this round trip is inefficient compared to storing electricity in batteries. Its smallness also makes hydrogen difficult to contain, so storing and transporting hydrogen is tricky and requires new infrastructure. Therefore, in any situation where it is feasible, electric batteries are preferable, and our first priority should be to improve battery technology and increase the supply and distribution of renewable energy. In the interim cases where batteries aren't feasible due to size or other constraints, hydrogen is a potential alternative.
Hydrogen is also necessary as the actual feedstock for industrial processes and products. Today, the production of hydrogen accounts for 6% of energy demand across industrial applications. By far the largest of these applications is the production of ammonia (used in fertilizer and explosives, among other things), followed by the production of methanol (ultimately used in everything from adhesives to windshield wiper fluid). In the future, hydrogen will also be a vital ingredient in the decarbonization of steel, a carbon-intensive industry projected to double by 2050.
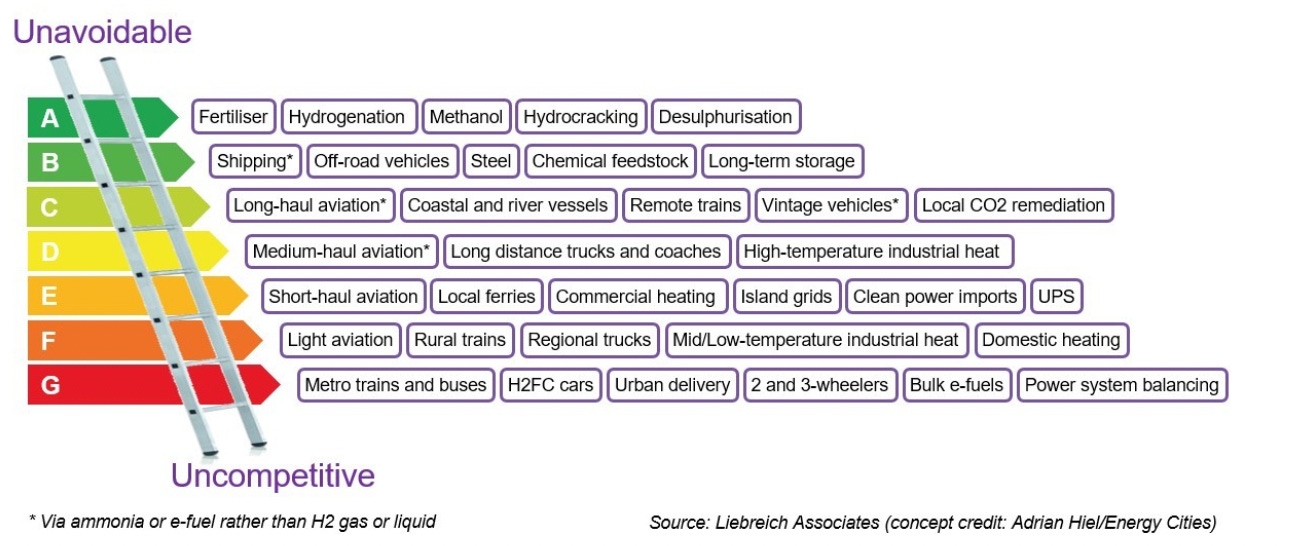
The below graphic is an excellent summary by Michael Liebreich of where hydrogen is a competitive (or in some cases, the sole) solution, and where it is not:
Importantly, all of the above assumes that the hydrogen supply is clean, or in the local parlance, "green." Today, there are several methods (and levels of cleanliness) for producing hydrogen, described in a rainbow of colors. Once the hydrogen is produced, it needs to be stored, transported to demand sources (industrial plants, fueling centers, etc.), and turned into electricity through a fuel cell or used as feedstock.
Gray and brown: Produced from natural gas and coal respectively, and emit CO2.
Blue: Produced in the same way as gray and brown, but uses carbon capture, utilization, and storage (CCUS) technologies to mitigate emissions. Blue hydrogen is controversial. Detractors question its true impact on greenhouse gases and accuse it of being a political chess move to prop up fossil fuel industries. Proponents call it a necessary transitional stepping stone in the race against climate change.
Green: Produced through electrolysis of water. When the electricity used in the process is renewable, green hydrogen is close to zero-emission across its life cycle.
Turquoise: An emerging technology that uses natural gas like gray and blue hydrogen, but creates a valuable form of solid carbon called "carbon black" as a byproduct (which can be stored or used in products like tires) instead of carbon dioxide.
While green or turquoise hydrogen is vital to decarbonizing heavy transportation and industry, it is not currently cost-effective compared to the dirtier colors. To close this "green premium," the cost of renewable electricity and electrolyzers would have to decrease, and the cost of carbon would have to increase.
Source: IEA (https://iea.blob.core.windows.net/assets/5bd46d7b-906a-4429-abda-e9c507a62341/GlobalHydrogenReview2021.pdf)
Key Players
Jobs in hydrogen can be split into "supply" and "demand." A nonexhaustive list of supply-side functions include:
Manufacturing electrolyzers for green hydrogen (Electric Hydrogen, ITM Power, Sunfire)
Producing turquoise hydrogen (C-Zero, Monolith Materials)
Integrating hydrogen plants with renewable energy sources (Aquaventus)
On the demand side, the opportunities to work with hydrogen span a startlingly diverse array of sectors:
Manufacturing fuel cells (Plug Power, cellcentric)
Creating electrofuels for heavy transportation (Infinium)
Manufacturing aviation (Universal Hydrogen, ZeroAvia)
Decarbonizing steel (H2 Green Steel, HYBRIT)
For the finance folks (that's me!), hydrogen funds include AP Ventures, HydrogenOne Capital, and FiveT Hydrogen.
What are the Challenges?
Greening the blue. A recent study from Robert Howarth and Mark Z. Jacobson found that the life cycle emissions of blue hydrogen are >20% greater than simply burning natural gas or coal. Emissions included methane leakage during natural gas extraction and the energy required to power the CCUS technology. Removing blue hydrogen from the equation could significantly undermine the role of hydrogen in future decarbonization, unless cleaner production methods are found. Turquoise hydrogen is currently a promising, if nascent, solution.
Making it cheaper. The electrolyzer technology key to green hydrogen is "fully mature," according to Jigar Shah, the founder of Generate Capital, but manufacturing needs to be scaled to make it truly cost-competitive. How can we help efficient technology get more so, while rapidly improving the unit economics, like what happened to solar over the last decade? How can we get the hydrogen electrolyzer to be the next photovoltaic cell?
Avoiding a Hindenburg. As illustrated by the Hindenburg disaster, hydrogen, despite its many merits, has a bad reputation for being highly flammable. As hydrogen expands beyond controlled steel and ammonia plants to enter our vehicles or our grid, we need stringent regulations in place to ensure that a high-vis accident doesn't sour public opinion and policymakers on what is a sorely needed solution (nuclear, anyone?).
🥳 What did you think? Let us know here.
Sources
https://hydrogencouncil.com/wp-content/uploads/2021/02/Hydrogen-Insights-2021-Report.pdf
https://www.newyorker.com/news/annals-of-a-warming-planet/the-promise-of-carbon-neutral-steel
https://www.linkedin.com/pulse/clean-hydrogen-ladder-v40-michael-liebreich/
https://spectra.mhi.com/achieving-net-zero-what-is-turquoise-hydrogen
https://www.nytimes.com/2021/08/12/climate/hydrogen-fuel-natural-gas-pollution.html
https://climatetechvc.org/🌍-hydrogen-hype-and-truth-with-jigar-shah/